Abstract
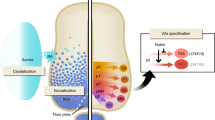
Forebrain γ-aminobutyric acid (GABA) interneurons have crucial roles in high-order brain function via modulating network activities and plasticity, and they are implicated in many psychiatric disorders. Availability of enriched functional human forebrain GABA interneurons, especially those from people affected by GABA interneuron deficit disease, will be instrumental to the investigation of disease pathogenesis and development of therapeutics. We describe a protocol for directed differentiation of forebrain GABA interneurons from human embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs) in a chemically defined system. In this protocol, human PSCs are first induced to primitive neuroepithelial cells over 10 d, and then patterned to NKX2.1-expressing medial ganglionic eminence progenitors by simple treatment with sonic hedgehog or its agonist purmorphamine over the next 2 weeks. These progenitors generate a nearly pure population of forebrain GABA interneurons by the sixth week. This simple and efficient protocol does not require transgenic modification or cell sorting, and it has been replicated with multiple human ESC and iPSC lines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Le Magueresse, C. & Monyer, H. GABAergic interneurons shape the functional maturation of the cortex. Neuron 77, 388–405 (2013).
Goulburn, A.L., Stanley, E.G., Elefanty, A.G. & Anderson, S.A. Generating GABAergic cerebral cortical interneurons from mouse and human embryonic stem cells. Stem Cell Res. 8, 416–426 (2012).
Nobrega-Pereira, S. et al. Postmitotic Nkx2-1 controls the migration of telencephalic interneurons by direct repression of guidance receptors. Neuron 59, 733–745 (2008).
Fishell, G. & Rudy, B. Mechanisms of inhibition within the telencephalon: “where the wild things are”. Annu. Rev. Neurosci. 34, 535–567 (2011).
Kriegstein, A.R. & Noctor, S.C. Patterns of neuronal migration in the embryonic cortex. Trends Neurosci. 27, 392–399 (2004).
Marin, O. & Rubenstein, J.L. Cell migration in the forebrain. Annu. Rev. Neurosci. 26, 441–483 (2003).
Letinic, K., Zoncu, R. & Rakic, P. Origin of GABAergic neurons in the human neocortex. Nature 417, 645–649 (2002).
Jakovcevski, I., Mayer, N. & Zecevic, N. Multiple origins of human neocortical interneurons are supported by distinct expression of transcription factors. Cereb. Cortex 21, 1771–1782 (2011).
Sussel, L., Marin, O., Kimura, S. & Rubenstein, J.L. Loss of Nkx2.1 homeobox gene function results in a ventral to dorsal molecular respecification within the basal telencephalon: evidence for a transformation of the pallidum into the striatum. Development 126, 3359–3370 (1999).
Danjo, T. et al. Subregional specification of embryonic stem cell-derived ventral telencephalic tissues by timed and combinatory treatment with extrinsic signals. J. Neurosci. 31, 1919–1933 (2011).
Campbell, K. Dorsal-ventral patterning in the mammalian telencephalon. Curr. Opin. Neurobiol. 13, 50–56 (2003).
Olsson, M., Campbell, K., Wictorin, K. & Bjorklund, A. Projection neurons in fetal striatal transplants are predominantly derived from the lateral ganglionic eminence. Neuroscience 69, 1169–1182 (1995).
Ma, L. et al. Human embryonic stem cell–derived GABA neurons correct locomotion deficits in quinolinic acid–lesioned mice. Cell Stem Cell 10, 455–464 (2012).
Zhao, Y. et al. The LIM-homeobox gene Lhx8 is required for the development of many cholinergic neurons in the mouse forebrain. Proc. Natl. Acad. Sci. USA 100, 9005–9010 (2003).
Flandin, P. et al. Lhx6 and Lhx8 coordinately induce neuronal expression of Shh that controls the generation of interneuron progenitors. Neuron 70, 939–950 (2011).
Manabe, T. et al. L3/Lhx8 is involved in the determination of cholinergic or GABAergic cell fate. J. Neurochem. 94, 723–730 (2005).
Maroof, A.M. et al. Directed differentiation and functional maturation of cortical interneurons from human embryonic stem cells. Cell Stem Cell 12, 559–572 (2013).
Nicholas, C.R. et al. Functional maturation of hPSC-derived forebrain interneurons requires an extended timeline and mimics human neural development. Cell Stem Cell 12, 573–586 (2013).
Liu, Y. et al. Medial ganglionic eminence-like cells derived from human embryonic stem cells correct learning and memory deficits. Nat. Biotechnol. 31, 440–447 (2013).
Li, X.J. et al. Directed differentiation of ventral spinal progenitors and motor neurons from human embryonic stem cells by small molecules. Stem Cells 26, 886–893 (2008).
Hu, B.Y. & Zhang, S.C. Differentiation of spinal motor neurons from pluripotent human stem cells. Nat. Protoc. 4, 1295–1304 (2009).
Li, Y. et al. Regulation of TrkA and ChAT expression in developing rat basal forebrain: evidence that both exogenous and endogenous NGF regulate differentiation of cholinergic neurons. J. Neurosci. 15, 2888–2905 (1995).
Zhang, S.C., Wernig, M., Duncan, I.D., Brustle, O. & Thomson, J.A. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat. Biotechnol. 19, 1129–1133 (2001).
Zhang, X. et al. Pax6 is a human neuroectoderm cell fate determinant. Cell Stem Cell 7, 90–100 (2010).
Pankratz, M.T. et al. Directed neural differentiation of human embryonic stem cells via an obligated primitive anterior stage. Stem Cells 25, 1511–1520 (2007).
Chambers, S.M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat. Biotechnol. 27, 275–280 (2009).
Cooper, O. et al. Differentiation of human ES and Parkinson's disease iPS cells into ventral midbrain dopaminergic neurons requires a high activity form of SHH, FGF8a and specific regionalization by retinoic acid. Mol. Cell Neurosci. 45, 258–266 (2010).
LaVaute, T.M. et al. Regulation of neural specification from human embryonic stem cells by BMP and FGF. Stem Cells 27, 1741–1749 (2009).
Reilly, J.O., Karavanova, I.D., Williams, K.P., Mahanthappa, N.K. & Allendoerfer, K.L. Cooperative effects of sonic hedgehog and NGF on basal forebrain cholinergic neurons. Mol. Cell Neurosci. 19, 88–96 (2002).
Krencik, R. & Zhang, S.C. Directed differentiation of functional astroglial subtypes from human pluripotent stem cells. Nat. Protoc. 6, 1710–1717 (2011).
Ogura, A., Morizane, A., Nakajima, Y., Miyamoto, S. & Takahashi, J. Gamma-secretase inhibitors prevent overgrowth of transplanted neural progenitors derived from human-induced pluripotent stem cells. Stem Cells Dev. 22, 374–382 (2013).
Acknowledgements
This study was supported in part by the US National Institutes of Health (NS045926, MH099587, NS076352), the Busta Family Foundation, the Bleser Family Foundation and the US National Institute of Child Health and Human Development (P30 HD03352).
Author information
Authors and Affiliations
Contributions
All authors contributed to the design of the experiments, analysis of data and writing of this paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Liu, Y., Liu, H., Sauvey, C. et al. Directed differentiation of forebrain GABA interneurons from human pluripotent stem cells. Nat Protoc 8, 1670–1679 (2013). https://doi.org/10.1038/nprot.2013.106
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.106
This article is cited by
-
Advancing preclinical models of psychiatric disorders with human brain organoid cultures
Molecular Psychiatry (2023)
-
Human organoids in basic research and clinical applications
Signal Transduction and Targeted Therapy (2022)
-
Routine culture and study of adult human brain cells from neurosurgical specimens
Nature Protocols (2022)
-
Induction of synapse formation by de novo neurotransmitter synthesis
Nature Communications (2022)
-
Grafted hPSC-derived GABA-ergic interneurons regulate seizures and specific cognitive function in temporal lobe epilepsy
npj Regenerative Medicine (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.