Abstract
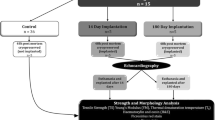
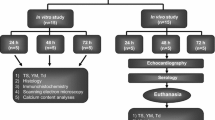
Homograft availability and durability remain big challenges. Increasing the post-mortem ischaemic harvesting time beyond 24 h increases the potential donor pool. Cryopreservation, routinely used to preserve homografts, damages the extracellular matrix (ECM), contributing to valve degeneration. Decellularization might preserve the ECM, promoting host-cell infiltration and contributing towards better clinical outcomes. This study compared the performance of cryopreserved versus decellularized pulmonary homografts in the right ventricle outflow tract (RVOT) of a juvenile ovine model. Homografts (n = 10) were harvested from juvenile sheep, subjected to 48 h post-mortem cold ischaemia, cryopreserved or decellularized and implanted in the RVOT of juvenile sheep for 180 days. Valve performance was monitored echocardiographically. Explanted leaflet and wall tissue evaluated histologically, on electron microscopical appearance, mechanical properties and calcium content. In both groups the annulus diameter increased. Cryopreserved homografts developed significant (¾) pulmonary regurgitation, with trivial regurgitation (¼) in the decellularized group. Macroscopically, explanted cryopreserved valve leaflets retracted and thickened while decellularized leaflets remained thin and pliable with good coaptation. Cryopreserved leaflets and walls demonstrated loss of interstitial cells with collapsed collagen, and decellularized scaffolds extensive, uniform ingrowth of host-cells with an intact collagen network. Calcific deposits were shown only in leaflets and walls of cryopreserved explants. Young fibroblasts, with vacuoles and rough endoplasmic reticulum in the cytoplasm, repopulated the leaflets and walls of decellularized scaffolds. Young’s modulus of wall tissue in both groups increased significantly. Cryopreserved valves deteriorate over time due to loss of cellularity and calcification, while decellularized scaffolds demonstrated host-cell repopulation, structural maintenance, tissue remodelling and growth potential.







Similar content being viewed by others
Data Availability
The manuscript has no associated data.
References
Ali ML, Kumar SP, Bjornstad K, Duran CM (1996) The sheep as an animal model for heart valve research. Cardiovasc Surg 4(4):543–549. https://doi.org/10.1177/096721099600400426
Armiger LC (1998) Postimplantation leaflet cellularity of valve allografts: are donor cells beneficial or detrimental? Ann Thorac Surg 66(6 Suppl):S233–S235. https://doi.org/10.1016/S0003-4975(98)00984-9
Bakhach J (2009) The cryopreservation of composite tissues: principles and recent advancement on cryopreservation of different type of tissues. Organogenesis 5(3):119–126. https://doi.org/10.4161/org.5.3.9583
Bancroft JD, Stevens A (1990) Theory and practice of histological techniques, 3rd edn. Churchill Livingstone, Edinburgh
Bester D, Botes L, van den Heever JJ, Kotze H, Dohmen P, Pomar JL, Smit FE (2018) Cadaver donation: structural integrity of pulmonary homografts harvested 48 h post mortem in the juvenile ovine model. Cell Tissue Bank 19(4):743–754. https://doi.org/10.1007/s10561-018-9729-7
Bester D, Smit FE, van den Heever JJ, Botes, L, Dohmen PMCE (2017) Detoxification and stabilization of implantable or transplantable biological material. EU. 16792990.0–1455
Bibevski S, Ruzmetov M, Fortuna RS, Turrentine MW, Brown JW, Ohye RG (2017) Performance of synergraft decellularized pulmonary allografts compared with standard cryopreserved allografts: results from multiinstitutional data. Ann Thorac Surg 103(3):869–874. https://doi.org/10.1016/j.athoracsur.2016.07.068
Biermann AC, Marzi J, Brauchle E, Wichmann JL, Arendt CT, Puntmann V, Nagel E, Abdelaziz S, Winter AG, Brockbank KGM, Layland S, Schenke-Layland K, Stock UA (2019) Improved long-term durability of allogeneic heart valves in the orthotopic sheep model. Eur J Cardiothorac Surg 55(3):484–493. https://doi.org/10.1093/ejcts/ezy292
Blum KM, Drews JD, Breuer CK (2018) Tissue-engineered heart valves: a call for mechanistic studies. Tissue Eng, Part B: Rev 24(3):240–253. https://doi.org/10.1089/ten.teb.2017.0425
Boethig D, Horke A, Hazekamp M, Meyns B, Rega F, Van Puyvelde J, Hübler M, Schmiady M, Ciubotaru A, Stellin G, Padalino M, Tsang V, Jashari R, Bobylev D, Tudorache I, Cebotari S, Haverich A, Sarikouch S (2019) A European study on decellularized homografts for pulmonary valve replacement: initial results from the prospective ESPOIR Trial and ESPOIR Registry data†. Eur J Cardiothorac Surg 56(3):503–509. https://doi.org/10.1093/ejcts/ezz054
Botes L, van den Heever JJ, Smit FE, Neethling WML (2012) Cardiac allografts: a 24-year South African experience. Cell Tissue Bank 13(1):139–146. https://doi.org/10.1007/s10561-010-9237-x
Bourgine PE, Pippenger BE, Todorov A, Tchang L, Martin I (2013) Tissue decellularization by activation of programmed cell death. Biomaterials 34(26):6099–6108. https://doi.org/10.1016/j.biomaterials.2013.04.058
Brockbank KG, Lightfoot FG, Song YC, Taylor MJ (2000) Interstitial ice formation in cryopreserved homografts: a possible cause of tissue deterioration and calcification in vivo. J Heart Valve Dis 9(2):200–206
Brown JW, Elkins RC, Clarke DR, Tweddell JS, Huddleston CB, Doty JR, Fehrenbacher JW, Takkenberg JJM (2010) Performance of the CryoValve SG human decellularized pulmonary valve in 342 patients relative to the conventional CryoValve at a mean follow-up of four years. J Thorac Cardiovasc Surg 139(2):339–348. https://doi.org/10.1016/j.jtcvs.2009.04.065
Canty EG, Kadler KE (2005) Procollagen trafficking, processing and fibrillogenesis. J Cell Sci 118(7):1341–1353. https://doi.org/10.1242/jcs.01731
Carr-White GS, Kilner PJ, Hon JK, Rutledge T, Edwards S, Burman ED, Pennell DJ, Yacoub MH (2001) Incidence, location, pathology, and significance of pulmonary homograft stenosis after the ross operation. Circulation 104(Suppl 1):16–20. https://doi.org/10.1161/hc37t1.094545
Crapo PM, Gilbert TW, Badylak SF (2011) An overview of tissue and whole organ decellularization processes. Biomaterials 32(12):3233–3243. https://doi.org/10.1016/j.biomaterials.2011.01.057
Cross PC, Mercer KL (1993) Cell and tissue ultrastructure – a functional perspective. WH Freeman, New York
da Costa FD, Costa AC, Prestes R, Domanski AC, Balbi EM, Ferreira AD, Lopes SV (2010) The early and midterm function of decellularized aortic valve allografts. Ann Thorac Surg 90(6):1854–1860. https://doi.org/10.1016/j.athoracsur.2010.08.022
da Dosta FD, Takkenberg JJ, Fornazari D, Balbi Filho EM, Colatusso C, Mokhles MM, da Costa AB, Sagrado AG, Ferreira AD, Fernandes T, Lopes SV (2014) Long-term results of the Ross operation: an 18-year single institutional experience. Eur J Cardiothorac Surg 46(3): 415–422. https://doi.org/10.1093/ejcts/ezu013
da Costa FDA, Dohmen PM, Lopes SV, Lacerda G, Pohl F, Vilani R, Affonso da Costa MB, Vieira ED, Yoschi S, Konertz W, Affonso da Costa I (2004) Comparison of cryopreserved homografts and decellularized porcine heterografts implanted in sheep. Artif Organs 28(4):366–370. https://doi.org/10.1111/j.1525-1594.2004.47357.x
Delmo Walter EM, de By TM, Meyer R, Hetzer R (2012) The future of heart valve banking and of homografts: perspective from the Deutsches Herzzentrum Berlin. HSR Proc Intensive Care Cardiovasc Anesth 4(2):97–108
Dijkman PE, Driessen-Mol A, Frese L, Hoerstrup SP, Baaijens FP (2012) Decellularized homologous tissue-engineered heart valves as off-the-shelf alternatives to xeno- and homografts. Biomaterials 33(18):4545–4554. https://doi.org/10.1016/j.biomaterials.2012.03.015
Dohmen PM, Ozaki S, Nitsch R, Yperman J, Flameng W, Konertz W (2003) A tissue engineered heart valve implanted in a juvenile sheep model. Med Sci Monit 9(4):97–104
Dohmen PM, da Costa F, Holinski S, Lopes SV, Yoshi S, Reichert LH, Villani R, Posner S, Konertz W (2006) Eur Surg Res 38(1):54–61. https://doi.org/10.1159/000091597
Elkins RC, Knott-Craig CJ, Ward KE, Mccue C, Lane MM (1994) Pulmonary autograft in children: realized growth potential. Ann Thorac Surg 57(6):1387–1393. https://doi.org/10.1016/0003-4975(94)90089-2
Erdbrügger W, Konertz W, Dohmen PM, Posner S, Ellerbrok H, Brodde OE, Robenek H, Modersohn D, Pruss A, Holinski S, Stein-Konertz M, Pauli G (2006) Decellularized xenogenic heart valves reveal remodeling and growth potential in vivo. Tissue Eng 12(8):2059–2068. https://doi.org/10.1089/ten.2006.12.2059
Fischlein T, Schütz A, Uhlig A, Frey R, Krupa W, Babic R, Thiery J, Reichart B (1994) Integrity and viability of homograft valves. Eur J Cardiothorac Surg 8(8):425–430. https://doi.org/10.1016/1010-7940(94)90084-1
Flameng W, Meuris B, Yperman J, de Visscher G, Herijgers P, Verbeken E (2006) Factors influencing calcification of cardiac bioprostheses in adolescent sheep. J Thorac Cardiovasc Surg 132(1):89–98. https://doi.org/10.1016/j.jtcvs.2006.02.036
Goecke T, Theodoridis K, Tudorache I, Ciubotaru A, Cebotari S, Ramm R, Hoffler K, Sarikouch S, Vasquez Rivera A, Haverich A, Wolkers WF, Hilfiker A (2018) In vivo performance of freeze-dried decellularized pulmonary heart valve allo- and xenografts orthotopically implanted into juvenile sheep. Acta Biomater 68:41–52. https://doi.org/10.1016/j.actbio.2017.11.041
Goffin YA, van Hoeck B, Jashari R, Soots G, Kalmar P (2000) Banking of cryopreserved heart valves in Europe: assessment of a 10-year operation in the European Homograft Bank (EHB). J Heart Valve Dis 9(2):207–214
Gomel MA, Lee R, Grande-Allen KJ (2019) Comparing the role of mechanical forces in vascular and valvular calcification progression. Front Cardiovasc Med 5:197. https://doi.org/10.3389/fcvm.2018.00197
Hennessy RS, Go JL, Hennessy RR, Tefft BJ, Jana S, Stoyles NJ, Al-Hijji MA, Thaden JJ, Pislaru SV, Simari RD, Stulak JM, Young MD, Lerman A (2017) Recellularization of a novel off-the-shelf valve following xenogenic implantation into the right ventricular outflow tract. PLoS ONE 12(8):e0181614–e0181614. https://doi.org/10.1371/journal.pone.0181614
Hodgson AN, Cross RHM, Bernard RTF (1990) An illustrated introduction to the ultrastructure of cells. Butterworths, Durban, South Africa
Hopkins RA, Jones AL, Wolfinbarger L, Moore MA, Bert AA, Lofland GK (2009) Decellularization reduces calcification while improving both durability and 1-year functional results of pulmonary homograft valves in juvenile sheep. J Thorac Cardiovasc Surg 137(4):907–913. https://doi.org/10.1016/j.jtcvs.2008.12.009
Jane-Wit D, Manes TD, Yi T, Qin L, Clark P, Kirkiles-Smith NC, Abrahimi P, Devalliere J, Moeckel G, Kulkarni S, Tellides G, Pober JS (2013) Alloantibody and complement promote T cell-mediated cardiac allograft vasculopathy through noncanonical nuclear factor-kappaB signaling in endothelial cells. Circulation 128(3):2504–2516. https://doi.org/10.1161/CIRCULATIONAHA.113.002972
Jashari R, Daenen W, Meyns B, Vanderkelen A (2004) Is ABO group incompatibility really the reason of accelerated failure of cryopreserved allografts in very young patients?–Echography assessment of the European Homograft Bank (EHB) cryopreserved allografts used for reconstruction of the right ventricular outflow tract. Cell Tissue Bank 5(4):253–259. https://doi.org/10.1007/s10561-004-1442-z
Laker L, Dohmen PM, Smit FE (2020) Synergy in a detergent combination results in superior decellularized bovine pericardial extracellular matrix scaffolds. J Biomed Mater Res, Part B: Appl Biomater 108(6):2571–2578. https://doi.org/10.1002/jbm.b.34588
Legare JF, Lee TD, Creaser K, Ross DB (2000) T lymphocytes mediate leaflet destruction and allograft aortic valve failure in rats. Ann Thorac Surg 70(4):1238–1245. https://doi.org/10.1016/S0003-4975(00)01677-5
Lehr EJ, Rayat GR, Chiu B, Churchill T, McGann LE, Coe JY, Ross DB (2011) Decellularization reduces immunogenicity of sheep pulmonary artery vascular patches. J Thorac Cardiovasc Surg 141(4):1056–1062. https://doi.org/10.1016/j.jtcvs.2010.02.060
Lisy M, Kalender G, Schenke-Layland K, Brockbank KG, Biermann A, Stock UA (2017) Allograft heart valves: current aspects and future applications. Biopreserv Biobank 15(2):148–157. https://doi.org/10.1089/bio.2016.0070
Manji RA, Zhu LF, Nijjar NK, Rayner DC, Korbutt GS, Churchill TA, Rajotte RV, Koshal A, Ross DB (2006) Glutaraldehyde-fixed bioprosthetic heart valve conduits calcify and fail from xenograft rejection. Circulation 114(4):318–327. https://doi.org/10.1161/CIRCULATIONAHA.105.549311
NIH (2011) Guide for the Care and Use of Laboratory Animals, 8th ed. Washington DC: National Academy of Sciences. https://www.ncbi.nlm.nih.gov/pubmed/21595115. Accessed 12 December 2019
Numata S, Fujisato T, Niwaya K, Ishibashi-Ueda H, Nakatani T, Kitamura S (2004) Immunological and histological evaluation of decellularized allograft in a pig model: comparison with cryopreserved allograft. J Heart Valve Dis 13(6):984–990
Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R (2018) Tissue-engineered grafts from human decellularized extracellular matrices: a systematic review and future perspectives. Int J Mol Sci. https://doi.org/10.3390/ijms19124117
Quinn RW, Hilbert SL, Bert AA, Drake BW, Bustamante JA, Fenton JE, Moriarty SJ, Neighbors SL, Lofland GK, Hopkins RA (2011) Performance and morphology of decellularized pulmonary valves implanted in juvenile sheep. Ann Thorac Surg 92(1):131–137. https://doi.org/10.1016/j.athoracsur.2011.03.039
Romeo JLR, Papageorgiou G, van de Woestijne PC, Takkenberg JJM, Westenberg LEH, van Beynum I, Bogers A, Mokhles MM (2018) Downsized cryopreserved and standard-sized allografts for right ventricular outflow tract reconstruction in children: long-term single-institutional experience. Interact Cardiovasc Thorac Surg 27(2):257–263. https://doi.org/10.1093/icvts/ivy057
Sarikouch S, Horke A, Tudorache I, Beerbaum P, Westhoff-Bleck M, Boethig D, Repin O, Maniuc L, Ciubotaru A, Haverich A, Cebotari S (2016) Decellularized fresh homografts for pulmonary valve replacement: a decade of clinical experience. Eur J Cardiothorac Surg 50(2):281–290. https://doi.org/10.1093/ejcts/ezw050
Sarikouch S, Theodoridis K, Hilfiker A, Boethig D, Laufer G, Andreas M, Cebotari S, Tudorache I, Bobylev D, Neubert L, Teiken K, Robertus JL, Jonigk D, Beerbaum P, Haverich A, Horke A (2019) Early insight into in vivo recellularization of cell-free allogenic heart valves. Ann Thorac Surg 108(2):581–589. https://doi.org/10.1016/j.athoracsur.2019.02.058
Schoen FJ (2008) Evolving concepts of cardiac valve dynamics: the continuum of development, functional structure, pathobiology, and tissue engineering. Circulation 118:1864–1880. https://doi.org/10.1161/CIRCULATIONAHA.108.805911
Selamet Tierney ES, Gersony WM, Altmann K, Solowiejczyk DE, Bevilacqua LM, Khan C, Krongrad E, Mosca RS, Quaegebeur JM, Apfel HD (2005) Pulmonary position cryopreserved homografts: durability in pediatric Ross and non-Ross patients. J Thorac Cardiovasc Surg 130(2):282–286. https://doi.org/10.1016/j.jtcvs.2005.04.003
Smit FE, Bester D, van den Heever JJ, Schlegel F, Botes L, Dohmen PM (2015) Does prolonged post-mortem cold ischaemic harvesting time influence cryopreserved pulmonary homograft tissue integrity? Cell Tissue Bank 16(4):531–544. https://doi.org/10.1007/s10561-015-9500-2
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26(1–2):31–43. https://doi.org/10.1016/S0022-5320(69)90033-1
Takkenberg JJ, van Herwerden LA, Eijkemans MJ, Bekkers JA, Bogers AJ (2002) Evolution of allograft aortic valve replacement over 13 years: results of 275 procedures. Eur J Cardiothorac Surg 21(4):683–691. https://doi.org/10.1016/S1010-7940(02)00025-8
Thubrikar MJ, Deck JD, Aouad J, Nolan SP (1983) Role of mechanical stress in calcification of aortic bioprosthetic valves. J Thorac Cardiovasc Surg 86(1):115–125. https://doi.org/10.1016/s0022-5223(19)39217-7
Tweddell JS, Pelech AN, Frommelt PC, Mussatto KA, Wyman JD, Fedderly RT, Berger S, Frommelt MA, Lewis DA, Friedberg DZ, Thomas JP, Sachdeva R, Litwin SB (2000) Factors affecting longevity of homograft valves used in right ventricular outflow tract reconstruction for congenital heart disease. Circulation. https://doi.org/10.1161/01.cir.102.suppl_3.iii-130
Vesely I, Casarotto DC, Gerosa G (2000) Mechanics of cryopreserved aortic and pulmonary homografts. J Heart Valve Dis 9(1):27–37
Zhang BL, Bianco RW, Schoen FJ (2019) Preclinical assessment of cardiac valve substitutes: current status and considerations for engineered tissue heart valves. Front Cardiovasc Med 6:72. https://doi.org/10.3389/fcvm.2019.00072
Acknowledgements
We would like to thank Miss H Grobler from the Centre for Electron Microscopy for all the tissue preparation for SEM and TEM, the UFS Animal Research Centre for their assistance, Department of Anaesthesiology and all perfusion technologists.
Funding
The study was funded by the Department of Cardiothoracic Surgery, University of the Free State, Bloemfontein, South Africa.
Author information
Authors and Affiliations
Contributions
All authors contributed to the article and deserve authorship.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
The interfaculty Animal Ethics Committee of the University of the Free State (UFS-AED2016/0101) approved the study.
Consent for publication
We certify that all authors have agreed to be listed in the manuscript when submitted to peer review journals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
van den Heever, J.J., Jordaan, C.J., Lewies, A. et al. Comparison of the function and structural integrity of cryopreserved pulmonary homografts versus decellularized pulmonary homografts after 180 days implantation in the juvenile ovine model. Cell Tissue Bank 23, 347–366 (2022). https://doi.org/10.1007/s10561-021-09948-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-021-09948-2